Protoreaction of Protoplasm
Vladimir
V. Matveev
Laboratory of Cell Physiology, Institute of Cytology, Russian Academy of
Sciences,
Tikhoretsky Ave
Cell
Mol Biol (Noisy-le-grand). 2005, 16;51(8):
715-723
Full
text [PDF]
Abstract.
My goal is to describe briefly the universal cellular reaction (UCR) to
external actions and agents. This general reaction was the main subject of
investigation by the scientific school of the outstanding Russian cytologist, Dmitrii Nasonov (1895-1957). The UCR consists of two phases
of complex changes in cellular viscosity and turbidity, in the cell's ability
to bind vital dyes, in the resting membrane potential, and in cellular
resistance to harmful actions. Works from the Nasonov School have shown that
these changes are based on structural-functional
transformations of many cell proteins that react uniformly to actions of
different physical and chemical nature. In general, these complex changes do
not depend on cell type, indicating the universal and ancient nature of the UCR
as well as its general biological significance. A new interpretation of the
mechanism of the universal reaction is proposed in
this paper, and a possible role for contractile proteins in the mechanism of
the UCR of muscle cells is presented. In addition, the concept of cell hydrophobicity is
introduced. Nasonov's School
proposed a concept of physiological standardization that allows comparison of
data obtained by different investigators and that will also
be described here.
Introduction.
According to an old Indian parable, well known in Russia, residents of the city
of blind people asked several respected citizens to act as experts and to
describe to them the nature of an elephant, about which they had heard much. It
happened that one of these animals was present near the walls of their city.
One expert who examined the elephant’s leg by feeling it came
to the conclusion that the elephant was a column. Another expert, upon
touching carefully the animal’s tail, stated that the elephant was a rope. The
expert who got the tusk was absolutely sure that the
elephant resembled a ploughshare. Clearly, the experts failed to agree and
continued to dispute all their lives, since each one felt that their case was based firmly on established facts. Thus, each of them
was in the right, but all of them were wrong on the whole.
Cell physiology and the scientists dealing with study of this discipline
somewhat remind us of the meaning of this parable. To some of them, cell
physiology focuses on the plasma membrane, to others the nucleus is the key,
yet others prefer seeing the key to the mysteries to be found
in signaling pathways. The “touching” of individual cell parts continues in
contemporary cell biology.
Fortunately, the cell itself gives us examples of its reactions that imply the
basis for generalizations, for a broad view of cell physiology. One such
example is the universal cellular reaction (UCR) to external actions, which was studied in detail by the physiological school of the
outstanding Russian scientist, Dmitrii Nasonov
(1895-1957), founder of the Institute of Cytology of the Russian Academy of
Sciences, and author of 117 publications including two monographs. At present,
the total number of publications from the Nasonov School is
estimated to be between 400 and 500. It is true that Nasonov himself
called this reaction unspecific, rather than universal. But
I consider the term “universal” to be more accurate and to better reflect the
physiological and biological significance of this reaction, and I will apply
this terminology here. The UCR is the uniform complex of substantial changes,
apparently occurring in all cell types, in response to external actions of all
kinds. The goal of this article is to describe some of these forgotten
investigations, and to consider them in terms of another paradigm, the
Association-Induction Hypothesis (14, 15) that seems to me to be a suitable basis
for such an analysis. The necessity to reinterpret the results of the Nasonov’s School and its heritage seems reasonable because
the corresponding literature, already old, can be found
to contain only the phenomenological
or quite general
accounts of the UCR. However, it seems to me that something better can be suggested in terms of contemporary biology. I hope
the reader will agree that, in the framework of this brief paper, only a schema
of this new approach to the problem can be presented.
I will consider this task completed if I manage to present to the reader at
least the general notion of the universal cellular reaction, and of its
possible mechanism.
A universal reaction of
the living cell. One of the least understood properties of the
living cell, apparently outside the scope of modern science, is its ability to
respond to stimuli of different
natures by the same
standard complex of structural and functional responses. It is upon this
phenomenon that the main efforts of Nasonov’s School were focused. In these studies
major attention was devoted to changes in cell properties, rather than to
descriptions of its steady states. A simple but quite efficient method to
investigate cell changes was to study binding of vital (non-toxic) dyes by
cells. This procedure became the key approach in studies by the School and was also accompanied by studies of such physical
characteristics as turbidity (transparency) of cytoplasm and nucleoplasm, their
viscosity, biopotentials, and resistance to damaging
actions by the agents discussed below.
The list of actions on the cell that were studied included: increased
temperature, mechanical stress, hydrostatic pressure, electric current, general
anesthetics, pH, medium tonicity, salts of heavy metals, hypoxia, and sound
irradiation (200-7000 Hz, 94 dB). These studies used epithelial, nerve, muscle,
connective tissue, the germ cells of various worms, echinoderms, coelenterates,
molluscs, crustaceans, insects, and other
invertebrates, as well as representatives of protozoa and some plant cells (see
20 for references).
Based on these abundant data, I present in Fig. 1 a universal complex of
cellular changes in response to the agents named above. It includes changes of
cell properties in the first phase and then in the second phase of both types
of responses.
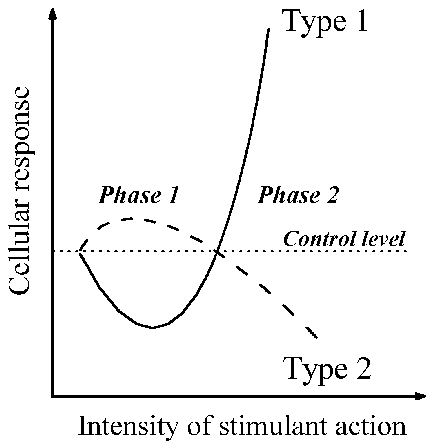
Fig. 1. Schematic presentation of the synchronous changes in cells that develop
during the course of the universal cell reaction, in response to actions of
various kinds. Changes in the cell’s turbidity and viscosity, and of its
ability to bind vital dyes, occur as described by type 1. Changes in cell
resistance to harmful agents, and of the resting membrane potential, occur as
in type 2. Further details are given in the text.
Changes of the first
type. Many works have established that changes in turbidity of the
cytoplasm and nucleus always occur in response to various actions on the cell.
The second phase of the reaction is easily observed
under the microscope: first the entire cell starts fluorescing with a pale blue
light, then white structures appear, and turbidity increases. These changes are
especially evident in nuclei, in which they appear even earlier than in
cytoplasm. During the first phase of the reaction, the transparency of the
protoplasm increases and, being a visual response, is best
recorded by instrumental methods. In this paper
the term “protoplasm” will be used, as it was in the days of Nasonov, to refer
to the entire living substance of cells. On the whole, these
changes can be characterized as follows: the size of intracellular colloids
initially decreases, and later, at the second phase of the reaction, begins to
increase, seemingly due to aggregation of the cytomatrix.
Another typical change characterizing the UCR response is an increase in
intracellular viscosity. Not infrequently, it becomes possible to record a
decrease of the viscosity (the first phase of type 1) before the beginning of
its increase (the second phase).
Nasonov’s School studied in the greatest
detail the ability of cells to bind vital dyes. At rest, the cell is almost never stained with vital dyes, and this is
especially true for the nucleus. However, under certain actions, the nucleus
and cytoplasm start adsorbing the dye intensively, and then dye adsorption
increases many times (up to 500% of the control or resting level, see Fig.1).
Especially intensively stained are the structures that are
found in the nucleus, such as chromatin granules, nucleolus and nuclear
envelope. In contrast it was found that during Phase 1
the ability of cells to bind dyes decreased by 10-30%. In both cases, the % values refer to the degree of dye binding by all of the
cells in the population studied.
Changes of the second type. Early in Nasonov’s career, great interest was
given to the data involved with the first phase of type 2 of the universal
reaction – namely, the increase in resistance of cells damaged by heat or
chemicals. This increase in resistance and stability was
manifested, in particular, by an increase in the ability of isolated
muscle to survive in Ringer’s solution. Such stabilization of muscle and other
cells was observed under the action of D2O, general
anesthetics and a variety of sugars, salts, vital dyes, and other compounds at
concentrations at which development of the UCR was delayed at the first phase. At a higher doses (concentrations) the increase in
resistance is replaced by its decrease during development of the second phase
of the UCR. In that case, the cells become much more sensitive to damaging
agents (see 28 for references).
Study by the School on the cellular resting membrane potential, recorded by
extra- and intracellular methods showed that membrane hyperpolarization
(relative to the resting state) took place during the early stages of
development of the reaction. Later, after a longer or more intensive action
(i.e. at the second phase) depolarization then begins (see 28 for references).
Such results can be added to other characteristics of
the UCR. For example, during the second phase an acidification of the nucleus
and cytoplasm occurs, as well as the release from the cell of various
substances including K+ along with the simultaneous influx of Na+
and Cl- (20).
It should be noted that the first phase of the UCR is
less intense and of shorter duration than the second phase, therefore, its
recording requires a high precision experiment.
A matter of principle importance should be especially
emphasized: the
universal reaction can develop not only in the cell as a whole, but also in its
individual parts, depending on the nature of the action. Hence, the
UCR can also be a localized
process. This peculiarity fascinated Nasonov and was always at the
center of his attention; he believed that there was no principal difference
between the localized reaction and the reaction of the whole cell in terms of
the spreading excitation of the action potential (20).
Finally, after cessation of a given action on the cell, all subsequent changes
show a reversed pattern and the cell gradually returns to the resting state. In
particular, dyes are released by the cell into the
surrounding solution against their concentration gradients during recovery. The
cytoplasm and nucleus revert to being colorless, and K+, various
phosphates and other substances that left the cell are now
taken up once more.
These changes can be summarized as follows: the first
phase of the UCR is characterized by an increase in cell stability, an elevated
resting membrane potential, and a decrease in cellular viscosity and turbidity,
as well as a slight decrease in the ability of the cell to bind vital dyes.
The second phase is characterized by a decrease of
cell stability and resting potential, a rise of viscosity and turbidity of the
protoplasm, and a significant increase in the ability of the cytoplasm and
nucleus to bind vital dyes.
Why protoreaction?
Experimental information accumulated over 40 years of investigations allowed
Nasonov to conclude that his universal cellular reaction is
based on reversible changes of cellular proteins (20). Indeed, changes
in protein solutions in
vitro are qualitatively similar to changes observed in living cells
under comparable conditions. Thus, proteins lose solubility and aggregate,
often with a rise in the viscosity of their solutions, and their ability to
bind dyes increases when stressed. On the other hand, the actions that increase
cell resistance also increase the stability of isolated proteins. Thus, agents
such as ethanol and chloral hydrate at a concentration at which they increase
resistance of the frog sartorius
muscle also increase stability of the glycerinated sartorius muscle
models (29), as well as of isolated actomyosin (16, 17). Those are important
and possibly profound observations.
The above changes in protein solutions are as universal as the UCR and they are induced by the actions of practically any physical or
chemical agent. The opposite is also true: thus, agents able to produce these
changes in proteins in vitro
also elicit the UCR (20). Comparing these many observations, the conclusion was easily reached that even the very first proteinoids (6) in evolution had the capability to produce
the universal reaction, and that has general biological significance (20). It
is in this context that I have referred to the universal reaction as the “protoreaction”,
as it is this response that must be the basis through evolution for the
formation of numerous regulatory systems in cells and, to a degree, will
continue to be reflected in physiological reactions in contemporary cells. But this term also has another meaning: in the protoreaction, we should find the fundamental processes
that must be responsible for the physical basis of life. So what is this
physical basis?
Physiological atom of
the living cell. Experts who consider that cell physiology is
very heavily influenced by membrane biology will hardly set about explaining
the mechanism of protoreaction, since we have already
stated that it takes place not only in whole cells, but also in local
intracellular areas as well as in membrane-deprived structures such as
glycerinated cell models and isolated proteins. For this reason, a promising
basis for analysis of protoreaction is, in my
opinion, Ling’s
Association-Induction Hypothesis (AIH) that has been
developed by its author for 4 decades and strives to be revolutionary, a
break-through in viewpoints on the cell based on its bulk-phase system (15,
19).
According to Ling’s
theory, the physical basis for life is an ion-water-protein complex – the
smallest structural unit that has the capability for protoreaction:
K+-H2O-PROTEINunf-ATP <–––> PROTEINf + H2O + ADP + Pi
+ K+,
where PROTEINunf represents unfolded
protein molecules, whose polypeptide chains are accessible to the solvent
water; where K+-, H2O-, ATP – represent protein-bound
potassium ions, water, and ATP; and PROTEINf
– the folded protein molecule, in which a significant part of the polypeptide
chain becomes inaccessible to water (see Fig. 44 in reference 15 for further
details).
The left part of this equation refers to a cell in the resting state, and the
right part to the state of activity or excitation. According to the AIH, it is such local changes that occur during action
potentials, muscle contractions, and other forms of cellular activity.
Transitions from the resting to the active are accompanied
by the release of free energy necessary to perform biological work (15).
Transitions between these two states of the ion-water-protein complex
represent, basically, a sol-gel transition or a cooperative phase transition.
According to this view, the triggering switches between these phases are what
generates the dynamics of life. These transitions are based
on regulated conformational protein changes that are not simply related to
shifts of atoms. It is probably more useful to evaluate relative conformational
changes by their accompanying thermodynamic changes rather than by values of
mechanistic shifts of parts of the molecules. If that approach is taken, the ion-water-protein complexes and their protoreaction are in essence the physiological “atoms” of
the cell in the sense that this is the minimal
structural entity able to produce the main interactions responsible for
cellular life, and its response to external disturbance. I suggest that the
living cell acts as if it is composed of such “atoms”, the various combinations
of which are then included into organelles, the cytomatrix
and various other cell structures. These “atoms” can acquire features of
specialization, but the main structural-functional principles of their activity
remain unchanged, so I will consider these “atoms” to be the basic units of the
living cell. Finally, I should note that not only the whole protein molecule
can act as a basic unit, but also that parts of it can operate that way. In
addition, when associations of such “atoms” take place with a high degree of
cooperativity, these “associations” or “complexes” can be regarded,
in some cases, as one “atom.”
This is the AIH logic, as I understand it. It seems to me that, on the whole, shifts of dynamic equilibrium between two
states of the basic unit reproduce, at the elementary
level, the protoreaction of cells, as
shown in Fig. 1. I suggest that these dynamics are the two states of a binary
code, upon which cell physiology operates.
Among other things I will later use the studies on dye
adsorption done by the Nasonov School to illustrate the basic features of the protoreaction. But first it is
necessary to examine a question not usually considered in this fashion: what is
the nature of cell hydrophobicity?
Cell hydrophobicity: a
missed role for proteins. For a long time, and up to the
present, the term hydrophobicity was mostly has been associated chiefly with
lipids. The well-known Meyer-Overton rule was always a strong argument in favor
of the lipid nature of biomembranes and of the
membrane theory of anesthesia. Until the 1960s, to be “hydrophobic” was
synonymous with being “lipid”, and the hydrophobic properties of the cell were
explained by the presence of its lipid membranes, first of
all, and primarily the plasma membrane. Indeed, based on these concepts,
numerous “lipid” theories of anesthesia were put
forward.
However, in the 1960s, when studying thermodynamic characteristics of the
thermodynamics of protein folding and unfolding, Brandts
(3) was the first to prove convincingly that during the folding of a protein
molecule, hydrophobic areas are formed internally which are inaccessible to
water. Initially the thermodynamics of conformational transitions in proteins
was the subject of study by a small group of specialists. However, with time,
it has become evident that hydrophobic areas within cells are
represented not only by lipids, as this was thought for more than 70
years, but also by proteins. The importance of this reappraisal is emphasized by the fact that, after water, protein is the
most abundant of all other constituents, comprising up to 65% of the dry mass
of cells, and greatly exceeds the total amount of lipid. What I propose here is
that the volume of the hydrophobic protein phase can greatly exceed that of the
hydrophobic lipid phase. However, I also recognize that the full significance
of this observation has not been understood and seemingly not
accepted by contemporary cell physiologists in terms of paradigms and working
hypotheses.
The next development essential in our understanding of cell hydrophobicity came
from the works of Katz and Simon (11) and Halsey et al. (9), who came to the
principally important conclusion that there was no difference between the physical properties
of hydrophobic sites of lipids and proteins as revealed by a thorough
thermodynamic analysis. In other words, hydrophobic compounds within cells will
interact with any
other hydrophobic site, regardless of location be it in proteins or in lipids.
This statement has an important consequence that will become clear when we
consider the example of valinomycin, a selective
potassium ionophore. It is accepted
as axiomatic
that this rather hydrophobic compound is dissolved only in the lipid phase of
the cell’s plasma membrane, and becomes a K+ carrier by virtue of
its concentration gradient. As a result and as repeatedly observed, cells
treated with valinomycin loses K+. This
“dogma” first appeared over 50 years ago when nothing was known about the
hydrophobic phase(s) in proteins, and still persists
to this day (10, 25). But we also know now that such
overly simplistic interpretations of valinomycin’s
effect on the cells are quite unacceptable. At present, it is evident that valinomycin can be inserted into any hydrophobic phase,
regardless of its nature, be that lipid or protein. Hence, valinomycin
can essentially change properties not only of membranes, but also of proteins
(including those of the cytomatrix); therefore, it is
no longer correct to explain the mechanism of action of this compound on the
cells only by
the action on changes in the permeability of the plasma membrane. Interestingly,
this statement, made on the basis of general
considerations, has become now been confirmed experimentally. It turned out
initially that valinomycin also had peculiar “side
effects”. Thus, it was revealed that valinomycin had
the ability to interact directly with cytochrome c oxidase (21, 26, 27), Ca2+-ATPase
(2), and (Ca2+,Mg2+)-ATPase of skeletal muscle
sarcoplasmic reticulum (5). It seems reasonable to suppose that other even
partially hydrophobic ionophores might also directly
interact with proteins. That topic seems worthy of further careful study.
Thus, after decades, it seems that the Meyer-Overton rule is neither a proof of
the lipid nature of membranes, nor evidence for the key role of membrane lipids
in anesthesiology. This rule merely indicates a role for hydrophobic
interactions in the cell permeability to the so-called lipophilic compounds.
The term “hydrophobic interaction” often is considered to be
synonymous with non-specificity. In reality, that term of hydrophobic
interactions is as non-informative about the degree of their specificity as is
the use of such terms as hydrogen bonds or ionic interactions. All these terms
merely indicate the physical nature of the interaction, rather than indicate
any degree of the level of their specificity. The latter quality depends on
numerous additional factors that are realized in the
microenvironments of the interacting molecules.
At present, the protein theory of anesthesia is commonly accepted, according to
which the targets of the anesthetic effect are hydrophobic sites located in
proteins (7), and this is of principal importance for the issues I consider in
this paper.
Phase transitions of
basic units and cell hydrophobicity. The evidence at my
disposal suggests that the basic unit protoreaction,
apart from other changes, leads to the appearance in the cell of a new physico-chemical
factor – hydrophobic areas formed by proteins. This statement is based on postulated properties of the basic units,
according to which a shift of the dynamic equilibrium between two states of the
basic unit (unfolded <–––> folded) to the right will bring about a
relative increase in the number of protein molecules in the folded state. This
will favor the formation of protein hydrophobic sites (areas, domains, pockets)
by virtue of the participation of hydrophobic side groups, both inside the
protein molecule and in intermolecular contacts (3). Thus, Ling’s model predicts that
at transition of the protoreaction into the second
phase of its development (see Fig. 1), the volume of the cellular protein
hydrophobic phase will increase. However, it is to be
stressed that Ling
does not consider such a possibility in his extensive writings (14, 15).
An increase of the hydrophobic phase volume fundamentally changes the
conditions of the intracellular environment and inevitably leads to a massive
redistribution of all
lipophilic compounds within the cell and between the cell and the external
medium. Such a redistribution should also involve key substances such as ATP,
since this compound is distinguished by significant
hydrophobicity (13). That seems to be a rather significant point with regard to
the UCR.
During Nasonov’s time, information on the properties
of proteins was scarce. It was cautiously believed by
his School that development of the protoreaction
leads to the appearance of additional fixed charges on proteins in cells, with
which vital dyes, known to be organic ions, presumably interacted. However, in
the review by Leo et al. (13) it is pointed out that
all vital dyes are characterized by high lipophilicity,
whereas the charge on these compounds produces no essential effect on their
hydrophobic interactions with other substances. This result is particularly
true for organic cations
(24). One of these organic cations, the vital dye neutral red, was widely
studied by the Nasonov School, and its use allowed them to obtain most of the
data on an increase of dye binding by the cell during the second phase of the protoreaction. Of great importance in this connection, is
the fact that neutral red is no different from general anesthetics (17) as far
as its mechanism of interaction with cell structures is concerned:
both the dye and general anesthetics interact with cell hydrophobic sites.
Thus, vital dyes are, in essence, indicators
of the volume of the cell hydrophobic phase formed by intracellular proteins.
Nasonov explained the increase in dye binding in the course of the protoreaction as being due to the “initial stage of protein
denaturation”, since proteins denaturated in vitro also bind dyes better than their native conformations. Both
in Nasonov’s works and in the context of the present
paper, use the term “denaturation” (i.e. loss of natural properties) seems
inappropriate, as it implies irreversible and probably lethal changes. In
discussions between Nasonov and his opponents, it was argued
that the cell is able to repair “denatured” proteins, and specifically those
with conformational modifications similar
to the denaturated state. However, from the point of
view of the above-considered dynamics of the basic unit states, restoration of
the cell to its initial state after protoreaction
looks not so much like reparation,
but more like the normal change of the basic unit states involved in mechanism
of UCR. Inappropriateness of the term “denaturation” was also indicated by
numerous data obtained by Nasonov and his colleagues, according to which the
normal functional activity of cells (secretion, muscle contraction, nerve
impulse propagation, transmission of synaptosome
signals, etc.) is also accompanied by an increase in the cellular viscosity,
turbidity and dye binding (see 20 for references).
Of great interest is the question of how vital dyes leave cells, against their concentration
gradients, after completion of the protoreaction and
a return of the cells to their resting state. First, it could be because a
transition to the resting state is accompanied by a
decrease in the volume of the hydrophobic phase (i.e. a decrease in the number
of the dye-binding hydrophobic centers). Second, according to the AIH, a large
fraction of cell water in the resting condition is in a state of restricted mobility
(“bound”) and is a poor solvent for large ions and various molecules (15). As a
result, these are excluded from intracellular water
into the surrounding solution. On the other hand, if we interpret the data
according to the membrane theory, it becomes necessary to postulate the
existence of active transport systems for each of the dyes studied by Nasonov’s School.
The concept of the basic unit helps explain as well the first phase of protoreaction when the ability of a cell to adsorb dyes is slightly reduced. The general explanation is based on the
assumption that the cell contains a small number of basic units in a folded
state under resting conditions since the balance “unfolded units <--->
folded units” is dynamic. If some influence on a cell leads to an even greater
displacement of the dynamic balance to the left, the total volume of a protein
hydrophobic phases in a cell will decrease in
comparison with the resting state. As a result, the cell’s ability
to bind lipophilic dyes will also decrease. For example, in the case of the
action of general anesthetics interacting hydrophobically
with cellular proteins, thermodynamic factors could play an important role. For
example, at a certain anesthetic concentration it could be advantageous
thermodynamically for protein hydrophobic side groups to make contact with the
mixed solvent (water + anesthetic) instead of with each other (3). As a result,
folded conformations of basic units, available in the resting state of a cell,
could become unstable and unfold, and expose its hydrophobic groups to the
mixed solvent. In that fashion, the dynamic balance between the two states of
the basic unit will be shifted to the left to a
greater degree than in the resting condition.
Another important factor in these processes is an increase of cellular ATP
during the first phase of the protoreaction (see 28
for references). According to the AIH, an increase in cellular ATP
concentration should lead to a shift to the left of the equilibrium between the
two states of the basic unit. Ling
(15) believes that ATP is the “cardinal adsorbent” and a key component of the
AIH. In the context of my paper an increase in ATP
concentration would strongly affect the dynamic equilibrium between the two
states of the basic unit: an increase in ATP concentration would shift the
equilibrium to the left, while a decrease would shift the equilibrium to the
right.
The significance of the increase in hydrophobicity of the cytoplasm and nucleus
for the functions of the cytoskeleton, signaling pathways, genome, and other
important cellular mechanisms remains virtually unknown and has yet to be investigated.
Intracellular viscosity.
I should first note that the studies done by the Nasonov School involved
descriptions of macroviscosity due to limitations in
the methodology of his era. Changes in the cytoskeleton are the first that come
to mind as an explanation for the changes in viscosity during the course of the
protoreaction. However, years of study on the effects
of anesthetics on cytoskeletal elements have shown that these compounds
depolymerize microtubules and microfilaments at clinical concentrations (1).
Thus, at the phase of cellular narcosis (i.e. at the second phase of the protoreaction) when the viscosity increases, this is
opposite to what would be expected from disassembly of the cytoskeleton.
Taking into account the basic unit properties, another explanation could
involve the bound state of intracellular K+. During tetanic
contraction of muscle and ethanol exposure, under conditions when the muscle
cell protoreaction reaches the second phase of its
development, K+ is known to leave the muscle due to K+
desorption from the K+-binding matrix (30). K+ efflux
from muscle during excitation is a well-known. In the
AIH context, free anionic groups on proteins produced by K+
desorption interact with fixed cationic groups on the same protein, or adjacent
ones. As a result of these interactions of fixed ions,
there appears a three-dimensional network of protein molecules bound to each
other in the cell, or in localized parts of it. This network is
believed to increase the viscosity significantly. A role in the
stabilization of such a network can also be played by interprotein hydrophobic interactions, where hydrophobic
side groups of adjacent protein molecules interact with each other, thereby
contributing to the stabilization of protein complexes or aggregates. Taking
into account the high protein concentration in cells, this “polymerization” of
basic units can proceed very fast, and involve large parts of cells or even
their entire volume. Such aggregations will inevitably lead to an elevation of
viscosity, an increase in the sizes of intracellular particles, and, hence, to
an increase of cell turbidity. Taking all this into account,
the cytoskeleton does not seem to play the key role in mechanisms underlying
the increase in viscosity.
Recall that, during the first phase of protoreaction,
the viscosity and turbidity fall below their resting levels. One can account
for those observations by a process involving the absorption. To do so, extra
anionic groups fixed to the basic unit are needed,
some of which come from sites that were previously occupied by other fixed
charges during the resting state. According to AIH logic, the number of fixed
anionic groups available for K+ binding increases when the cellular
ATP concentration rises. This theoretical prediction is in accord with the
above-mentioned data showing an increase in ATP during the first phase of the protoreaction (see 28 for references). Thus, an increase in
ATP synthesis and its excessive binding (compared with the resting state) by
basic units results in the breakage of an additional number of ionic bonds between proteins, and an
increase in the number of fixed anionic groups that can bind K+. It is further proposed that the above is accompanied by a
partial “depolymerization” of the three-dimensional
network of protein molecules, because some of the ionic bonds participating in
its stabilization are broken. Such a process of weakening of interprotein interactions would also be reflected as a
decrease in cell viscosity and an increase in its transparency as a result of the dissociation of protein aggregates.
Unfortunately, cell viscosity and K+ content, as far as I know, have always been studied separately. Consequently, one can
only refer to indirect evidence in favor of the above-described mechanism. Such indirect evidence comes from an interesting work by Troshina (31) showing that, under the action of insulin on
frog sartorius
muscle, the resting potential of the muscle fibers increases, while their
ability to adsorb neutral red decreases; hence, insulin produces the first
phase of the protoreaction in this muscle, during
which viscosity and turbidity of the sarcoplasm are known to decrease.
On the other hand, it is well established that insulin
increases the K+ content in muscle (4) which, according to the AIH,
could be due to the appearance of additional sites for K+ binding,
and to a corresponding decrease in stability of the protein matrix, as
discussed above. As a result, the dynamic equilibrium in the basic unit shifts
to the left to a greater degree than in the resting state, leading to a
decrease in viscosity and the ability to bind vital dyes.
It seems that the same effect can be produced by any
action that increases cellular ATP content since this increase is accompanied
by a rise in intracellular K+ content (8). In this connection, it is
interesting that these actions (classical for Nasonov’s
School) lead to an increase in creatine phosphate and
ATP in the cell, since these also increase cell resistance / stability (see 28
for references). Based on the above discussion, the following “rule” can be formulated: the greater the shift of dynamic
equilibrium between two states of basic units toward the left, the higher the
cell resistance and stability.
Thus, major cause of changes
in colloidal properties of cells, including rheological ones, seems to be assigned to the state of K+-binding by the
cellular matrix, the extent of which differs at different phases of the protoreaction.
Limiting proteins.
From the point of view of the AIH, basic units play the key role in maintaining
fundamental physico-chemical conditions of the
intracellular medium, which underlie the entire structural-functional
organization of cells. This gives good grounds to the belief that the loss by
basic units of the ability to perform their function would be sufficient for
cell death. If so, the proteins that are the structural basis of these units
can be called “limiting proteins” – those that play a critically important role
in providing the necessary conditions for metabolism and, therefore, life.
This theoretical anticipation has been confirmed
experimentally. Rosenberg et al. (23) studied the kinetic parameters of thermal
protein denaturation and thermal death of unicellular and multicellular
organisms. They came to the paradoxical conclusion that denaturation of one
protein, or of a small number of proteins with close properties (that the
authors called limiting
proteins) were sufficient for thermal death of a cell or organism.
From the point of view of the AIH, such proteins might be those of the K+-binding
cell matrix. An important question arises: what can be said
about the nature of these proteins?
As already noted here, the first protoreaction phase is characterized by an increase in cell resistance to
damaging factors, including thermal damage. For instance, in Ringer’s solution
containing 6 mM chloral hydrate or 680 mM ethanol the survival time of frog sartorius muscle is twice as long as that of control
preparations. Similar effects have also been obtained
using other chemical agents (see 28 for references). The question then is: which intracellular structures and/or proteins are the
targets of the actions responsible for an increase in resistance of the muscle
cell? Of course, there are many proteins in cells, and their properties differ
greatly. For instance, the maximal stabilizing effect of ethanol on ribonuclease is achieved at 2000
mM ethanol (3), whereas 680 mM is sufficient in the case of actomyosin (16).
In this connection, it is interesting to compare data obtained on living muscle and glycerol-treated muscle models
(see 28 for references), and on isolated actomyosin
(12, 16, 17). It has been established that stability
of all these preparations increased over the
same concentration range for chloral hydrate (maximum effect at 6
mM) and ethanol (maximum effect at 680 mM). In other words, this response of
the living cell is, to some degree, reproduced
by isolated proteins, specifically, by the contractile muscle proteins. This
astonishing observation merits more detailed study.
But why does actomyosin give such a good correlation
with living muscle in terms of these effects? Is this because of the high
content of these proteins in muscle? Or do the
contractile proteins play some additional key role in enabling viability of
muscle cells? One possible answer might be connected
to the fact that the contractile proteins bind the majority of K+
present in muscle (see 15 for references) and thereby are the structural
origins of the basic units of the UCR in muscle cells. If this is really so,
then the contractile proteins represent the K+-binding matrix, whose
stability is entirely responsible for cell viability. In that case, it is clear
that inactivation of the K+-binding matrix alone could make
functioning of muscle cells impossible. And, to the contrary, actions that
stabilize contractile proteins in
vivo also
make the treated muscle cell more resistant to malfunction. Apart from the key
role of contractile proteins as the basic units, they also play an important
role in the transmission of signals within muscle cells (18). Under such
circumstances, and in this context, contractile proteins can
indeed be considered limiting, and the above-mentioned experimental data
provide additional evidence in support of the conclusions of Rosenberg et al.
(23) about the cause of the thermal death of cells and multicellular organisms.
Protoreaction as a
physiological standard. It is easy to see that the protoreaction represents a non-linear response of cells to
some action. This means that the same stimulus can produce different results
depending on its intensity. This partly explains numerous controversies in the
literature, as authors studying some particular property of the cell do not
suspect that under their experimental conditions, the protoreaction
can develop, so that the cell properties being studied
depend essentially on the phase involved. Analysis of the results obtained,
without considering the physiological background under which they are obtained, is not likely to be correct. So it is important to know in which state of protoreaction cells are when they are being studied.
Indeed, it is very likely that protoreaction takes
place in every case if a cell is affected by any
method. One can only compare those effects that are developed
against a background of the same
phase of protoreaction (see Fig. 1)
according to the rule "all other conditions should be equal" (ceteris
paribus). In this way, numerous cell effects could be standardized, depending
on the protoreaction phase in which they were
observed.
In my opinion, the best indicator of the protoreaction
is a change in the hydrophobicity of cells or of certain intracellular
structures. Thus, an increase in nuclear hydrophobicity might initiate some
reactions, while preventing others. For example, it is very unlikely that
signalling systems in cells will operate similarly in the hydrophilic (Phase 1)
and hydrophobic (Phase 2) regions of cytoplasm or nucleus. All these issues are
extremely interesting and important to increase of effectiveness of science,
but are almost entirely uninvestigated.
Why can the protoreaction be used
as a standard? Because the entire body of scientific evidence accumulated by Nasonov’s School supports the claim, with some degree of
certainty, that the protoreaction is the only cell reaction that, in spite
of its complexity, has a universal and general biological character.
Furthermore, the complex changes occurring during development of the protoreaction appear in all cell types, at the scale of the
entire cell as well as intracellular structures, including molecular complexes.
The structural-functional principles that underlie the protoreaction
can be revealed in greatly different ways in the
nucleus, cytoplasm, organelles, during muscle contraction, nerve impulse
propagation, apoptosis, and so on, but the principles themselves remain
invariant.
Conclusion.
Currently, the ideas, approaches and methods of study developed by Nasonov’s School have essentially been
forgotten. But it is absolutely clear to those
who still remember this page of history of Russian science that the School studied
some fundamental cell properties, whose significance for biology is not
understood up to the present time. It is necessary to continue these
investigations of the Nasonov School, at the least because Nature never
disappoints those who study successively its fundamental manifestations. In my
view, one such manifestation is undoubtedly the protoreaction.
Here I have outlined merely the general scheme of the UCR / protoreaction
and its possible interpretation based on the Association Induction Hypothesis
of Gilbert Ling. It is
certainly evident that many aspects of this approach need further study and
experimental confirmation. But something else also
seems evident: only after carefully comparing the findings of the Nasonov
School with the main features of the AIH, which I tried to do here, does it
becomes clear as to which issues need further study. Formation of a plan of
investigation is one of the challenges of a good theory.
Acknowlegement. I am indebted to
James Clegg for critical comments on this article. I also wish to thank Leonid Pevzner and Denys Wheatley for their comments.
REFERENCES
1. Allison, A.C., The effects of inhalational anesthetics on proteins. In:
Molecular Mechanisms in General Anaesthesia, Halsey,
M.J., Millar, R.A. and Sutton, J.A. (eds), Churchill
Livingstone, Edinburg, London, New York, 1974, pp. 164-181.
2. Beeler, T.J. and Gable, K.S., Phosphate, nitrendipine
and valinomycin increase the Ca2+/ATP
coupling ratio of rat skeletal muscle sarcoplasmic reticulum Ca2+-ATPase.
Biochim. Biophys. Acta 1994, 1189: 189-194.
3. Brandts, J.F., Conformational transitions of
proteins in water and in aqueous mixtures. In: Structure and Stability of
Biological Macromolecules, Timasheft, S. N., and Fasman, G. D. (eds), Marcel
Dekker, Inc., New York, 1969, pp. 213–290.
4. Creese, R., D’Silva,
J.L. and Northover, J., Effect of insulin on sodium
in muscle. Nature 1958, 181: 1278.
5. Davidson, G.A. and Berman, M.C., Interaction of valinomycin
and monovalent cations with the Ca2+,Mg2+-ATPase of
skeletal muscle sarcoplasmic reticulum. J. Biol. Chem. 1985, 260: 7325-7329.
6. Fox, S.W., A
theory of macromolecular and cellular origins. Nature 1965, 205: 328-340.
7. Franks, N.P. and Lieb, W.R., Seeing the light: protein
theories of general anesthesia. Anesthesiology 2004, 101: 235-237.
8. Gulati, J., Ochesenfeld, M.M. and Ling, G.N., Metabolic
cooperative control of electrolyte levels by adenosine triphosphate in the frog
muscle. Biophys. J. 1971, 11: 973-980.
9. Halsey, M.J., Brown, F.F. and Richards, R.E., Perturbations
of model protein systems as a basis for central and peripheral mechanisms of
general anaesthesia. In: Molecular interactions
and activity in proteins. Porter, R. and Fitzsimons, D.W. (eds), Excerpta Medica, Amsterdam, Oxford, New York, 1978, pp. 123-132.
10. Inai, Y., Yabuki, M., Kanno, T., Akiyama, J., Yasuda, T. and Utsumi,
K. Valinomycin induces apoptosis of ascites hepatoma
cells (AH-130) in relation to mitochondrial membrane potential. Cell Struct. Funct. 1997,
22: 555-563.
11. Katz, Y. and Simon, S.A., Physical
parameters of the anesthetic site. Biochim. Biophys. Acta 1977, 471: 1-15.
12. Kovaleva, T.A., Suzdalskaya,
I.P., Butiagina, N.V., Stepanova,
T.P. and Gnetov, A.V., Enhancing the temperature
stability of muscle proteins under the action of chloral hydrate and urea. Fiziol. Zh. SSSR Im. I. M. Sechenova 1991, 77:
159-162. (In Russian)
13. Leo, A., Hansch, C. and Elkins, D., Partition
coefficients and their use. Chem. Rev. 1971, 71: 525-616.
14. Ling, G.N.,
A
Physical Theory of the Living State: the Association-Induction Hypothesis, Blaisdell Publ. Co., Waltham, Mass., 1962.
15. Ling, G.N.,
Life
at the Cell and Below-Cell Level. The Hidden History of a Fundamental
Revolution in Biology, Pacific Press, New York, 2001.
16. Matveev, V.V., Denaturation time of actomyosin exposed to different
chemicals. Tsitologiia 1975, 17: 1278-1282. (In
Russian)
17. Matveev, V.V., Enhanced stability of isolated muscles and actomyosin due to
action of anesthetics at subnarcotic concentrations. Tsitologiia 1987, 29: 197-201. (In Russian)
18. Matveev, V.V., Evidence of a new type of protein-protein interaction:
desensitized actomyosin blocks Ca2+-sensitivity of the natural one.
A possible model for an intracellular signalling system related to actin
filaments. Physiol. Chem. Phys. Med. NMR 2000, 32: 167-178.
19. Matveev, V.V., Revolution and counter revolution in cell physiology. Cell Biol. Int. 2002, 26: 305–308.
20. Nasonov, D.N.,
Local
Reaction of Protoplasm and Gradual Excitation (English Transl. by Halpern,
Y.S.), National Science Foundation, available at Office of Technical Services,
US Department of Commerce, Washington, D.C., 1962.
The book in Chinese.
21. Nicholls, P. and He, J., Direct and indirect effects of valinomycin
upon cytochrome c oxidase. Arch. Biochem. Biophys. 1993, 301: 305-310.
22. Porter, J., Pickup, R. and Edwards, C., Membrane hyperpolarisation
by valinomycin and its limitations for bacterial
viability assessment using rhodamine 123 and flow cytometry. FEMS Microbiol Lett. 1995, 132: 259-262.
23. Rosenberg, B., Kemeny, G., Switzer, R.C. and
Hamilton, T.C., Quantitative
evidence for protein denaturation as the cause of thermal death. Nature
1971, 232: 471-473.
24. Roth, S, Seeman, P., The
membrane concentrations of neutral and positive anesthetics (alcohols,
chlorpromazine, morphine) fit the Meyer-Overton rule
of anesthesia; negative narcotics do not. Biochim.
Biophys. Acta. 1972, 255: 207-219.
25. Shapiro, H.M., Cell membrane potential analysis. Methods Cell Biol. 1994,
41: 121-133.
26. Steverding, D. and Kadenbach,
B., Valinomycin binds stoichiometrically
to cytochrome c oxidase and changes its structure and function. Biochem. Biophys. Res. Commun. 1989, 160: 1132-1139.
27. Steverding, D. and Kadenbach,
B., The K+-ionophores nonactin
and valinomycin interact differently with the protein
of reconstituted cytochrome c oxidase. J. Bioenerg. Biomembr. 1990, 22: 197-205.
28. Suzdalskaya, I.P., Increase in stability of
isolated tissues and proteins caused by action of chemical stimulants. Tsitologiia 1977, 19: 839-849. (In Russian)
29. Suzdalskaya, I.P., Kiro,
M.B., Increase in the time of conservation of contractility of glycerinated
muscles exposed to subthreshhold concentrations of
chemical agents. Tsitologiia 1975, 17: 50-54. (In
Russian)
30. Troshin, A.S.,
Problems
of Cell Permeability, (English Transl. by Hell, M.G. and Widdas, W.F.), Pergamon Press,
London, 1966. Edition of 1956 in
Russian and in Chinese:
PDF
DJVU
31. Troshina, V.P., Effect of insulin on natural
properties of the frog muscle. Tsitologiia 1964, 6:
751-754. (In Russian)
Full
text [PDF]
Web-Links
The
journal "Physiological
Chemistry and Physics and Medical NMR"
Pollack G.H. Water, Energy and Life. (Lecture) (Link 1)
(Link 2)